on weekdays from 10:00 to 18:00

on weekdays from 10:00 to 18:00

KardiRu2
The device for ECG registration, 4 electrodes, 6 leads. Transfer research via SIM card or WI-FI.
With KardiRu2 device you’ll be able:
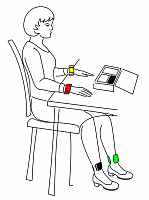
 Self-register ECG for yourself and family members anytime, anywhere.
Self-register ECG for yourself and family members anytime, anywhere.
 Get access to the entire ECG history through your personal account or mobile application.
Get access to the entire ECG history through your personal account or mobile application.
 Get an automatic ECG description.
Get an automatic ECG description.
 Receive an automatic assessment of the measurement in a traffic signal version.
Receive an automatic assessment of the measurement in a traffic signal version.
 Save, print and share ECG results.
Save, print and share ECG results.
 Get medical advisement if necessary.
Get medical advisement if necessary.
How it works
Reports
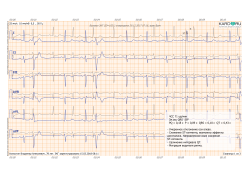
After each study, two reports are automatically generated, which are available in KardiRu personal account or in the mobile application.
1. ECG 6 leads with ECG description
https://www.kardi.ru/files/demo/KardiRuConclusion-ECG.pdf
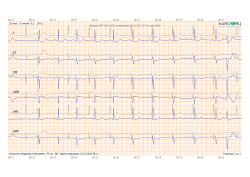
2. Additional ECG assessment methods and automatic conclusion
https://www.kardi.ru/files/demo/KardiRuConclusion-DK.pdf
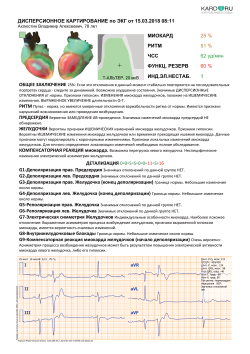
You can consult our doctors online by sending a measurement via your account, or show the measurement to your doctor.
Who needs a KardiRu2 device
 If you or your family members suffer from arrhythmias. You will have a device in an easily accessible place. You can always fix the attack of arrhythmia by measurement.
If you or your family members suffer from arrhythmias. You will have a device in an easily accessible place. You can always fix the attack of arrhythmia by measurement.
 Ordinary people to improve the quality of life in order to exclude factors that negatively affect the cardiovascular system. Through measurement, you will identify and exclude these factors.
Ordinary people to improve the quality of life in order to exclude factors that negatively affect the cardiovascular system. Through measurement, you will identify and exclude these factors.
 Amateur athletes to control the effectiveness of their workouts, to prevent overtraining.
Amateur athletes to control the effectiveness of their workouts, to prevent overtraining.
 Professional athletes to exclude accidents in overload mode.
Professional athletes to exclude accidents in overload mode.
 Patients after a heart attack, insult, heart surgery, after RF ablation.
Patients after a heart attack, insult, heart surgery, after RF ablation.
 Ordinary people over 40, who sometimes feel discomfort in the heart..
Ordinary people over 40, who sometimes feel discomfort in the heart..
 Children over 6 years old with congenital or acquired diseases of the cardiovascular system.
Children over 6 years old with congenital or acquired diseases of the cardiovascular system.
 Patients with coronary heart disease.
Patients with coronary heart disease.
 Patients who need remote consultation with a doctor from another region.
Patients who need remote consultation with a doctor from another region.
 Patients with diabetes to control the cardiovascular system.
Patients with diabetes to control the cardiovascular system.
 To control medication or drug therapy.
To control medication or drug therapy.
 For daily monitoring of the dynamics of the cardiovascular system.
For daily monitoring of the dynamics of the cardiovascular system.
 To control the QT interval and prevent life-threatening arrhythmias.
To control the QT interval and prevent life-threatening arrhythmias.
 For control and prevention of cardiomyopathies and myocarditis during recovery from acute respiratory viral infections, influenza and other infections.
For control and prevention of cardiomyopathies and myocarditis during recovery from acute respiratory viral infections, influenza and other infections.
Device features
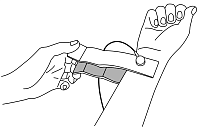
 6 standard ECG leads from 4 electrodes are recorded: I, II, III, aVR, aVL, aVF. Electrodes are put on hands and legs. To register a study, you do not need to undress.
6 standard ECG leads from 4 electrodes are recorded: I, II, III, aVR, aVL, aVF. Electrodes are put on hands and legs. To register a study, you do not need to undress.
 High quality of ECG registration (complies with GOST R IEC 60601-2-25)
High quality of ECG registration (complies with GOST R IEC 60601-2-25)
 The kit comes with quality reusable electrodes.
The kit comes with quality reusable electrodes.
 The duration of registration of the measurement is 30 seconds.
The duration of registration of the measurement is 30 seconds.
 It is possible to see the ECG at the time of registration.
It is possible to see the ECG at the time of registration.
 The duration of transmission of one measurement is from 3 to 6 minutes.
The duration of transmission of one measurement is from 3 to 6 minutes.
 To transmit the measurement, the Internet channel of the mobile operator is used.
To transmit the measurement, the Internet channel of the mobile operator is used.
 If the communication channel does not work, then the study is stored in the device.
If the communication channel does not work, then the study is stored in the device.
 When used, no medical or technical education is required. It is very easy to use the device for a patient of any age.
When used, no medical or technical education is required. It is very easy to use the device for a patient of any age.
 It is possible to print a measurement or send it online to your doctor.
It is possible to print a measurement or send it online to your doctor.
 The device is a medical product registered by Roszdravnadzor.
The device is a medical product registered by Roszdravnadzor.
MANUFACTURER AND ENABLING AND CERTIFYING DOCUMENTS
"The hardware and software suite for heart screening" KardiRu "Technical Notes 9442-038-17635079-2012" was tested and classified as fit for use.
| Medical device | The hardware and software suite for heart screening" KardiRu " |
|---|---|
| Product license | №RZN 2013/778 of June 28, 2013 Download the Product license |
| Certificate of Conformance | of December 26, 2016 Download the Certificate of Conformance |
| Offer Contract | of March 03, 2017 Download the Offer Contract |
| Manufacturer | LLC “Medical Computer Systems” |
| Manufacturer’s website | www.mks.ru |
| Classification | Class 2b, noninvasive, active device |
| Kind of medical device | 024947 The device for screening express heart state assessment of by ECG signals 215 04 02 13 |
| Complies with | Technical Notes 9442-038–17635079-2012, GOST R 50444-92, GOST R 50267.0-92, GOST R 50267.0.2-2005, GOST R 50267.0.4-99, GOST R 50267.25-94, GOST R IEC 60601-2-51-2008 |
Delivery
We understand that the ability to do ECG on their own at home is a necessity for many of our users.
Dispatch across Russia starts the next working day after ordering.
And we tightly control the delivery time of our devices for the customers.